BLD Insights
NHPI Esters for Decarboxylative Couplings
17 January 2023
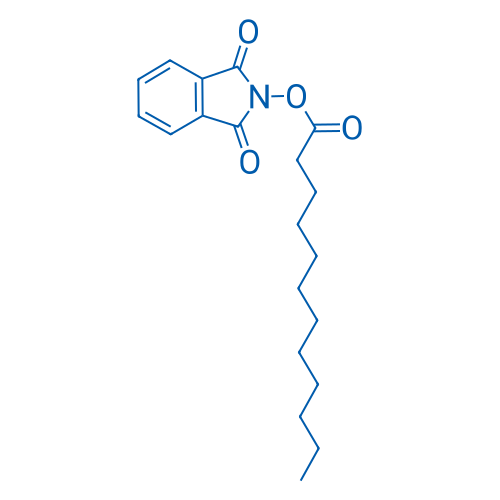
1,3-Dioxoisoindolin-2-yl dodecanoate
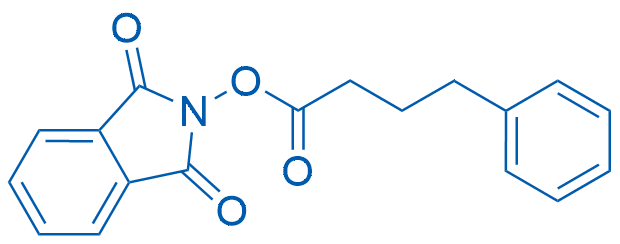
1,3-Dioxoisoindolin-2-yl 4-phenylbutanoate
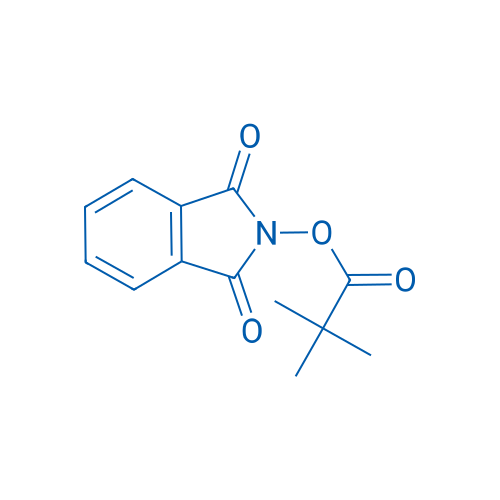
1,3-Dioxoisoindolin-2-yl pivalate
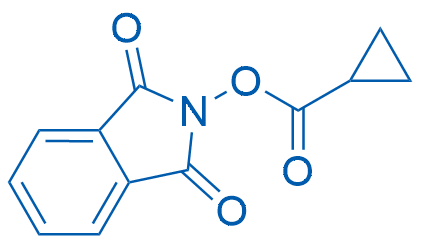
1,3-Dioxoisoindolin-2-yl cyclopropanecarboxylate
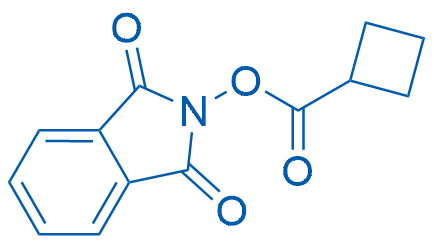
1,3-Dioxoisoindolin-2-yl cyclobutanecarboxylate
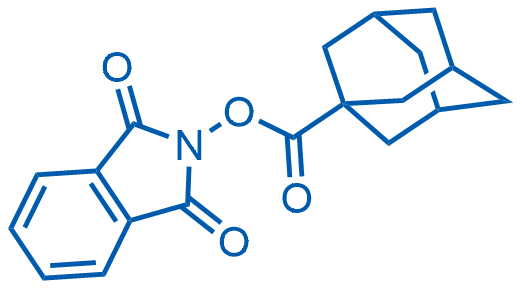
1,3-Dioxoisoindolin-2-yl adamantane-1-carboxylate
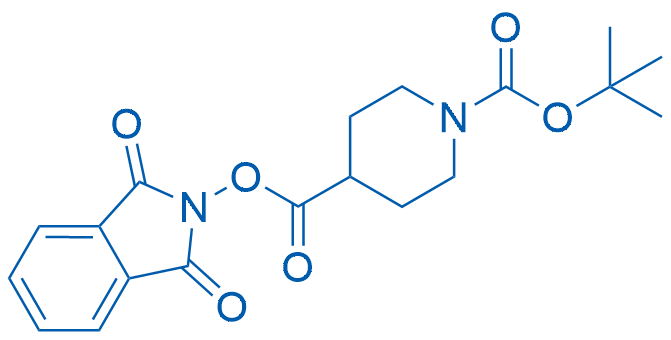
1-(tert-Butyl) 4-(1,3-dioxoisoindolin-2-yl) piperidine-1,4-dicarboxylate
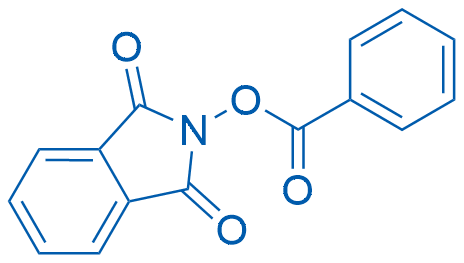
1,3-Dioxoisoindolin-2-yl benzoate
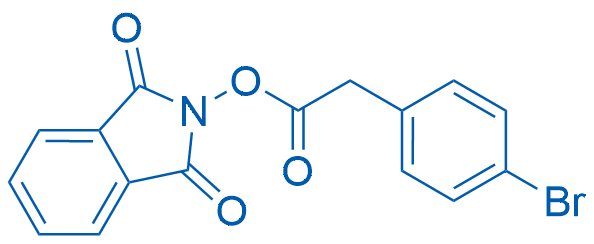
1,3-Dioxoisoindolin-2-yl 2-(4-bromophenyl)acetate
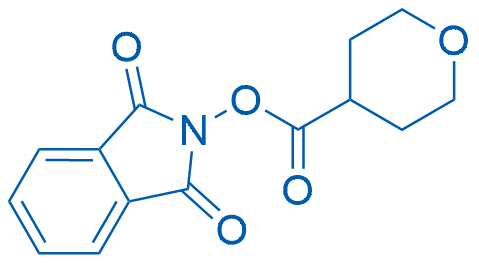
1,3-Dioxoisoindolin-2-yl tetrahydro-2H-pyran-4-carboxylate

1,3-Dioxoisoindolin-2-yl dodecanoate

1,3-Dioxoisoindolin-2-yl 4-phenylbutanoate

1,3-Dioxoisoindolin-2-yl pivalate

1,3-Dioxoisoindolin-2-yl cyclopropanecarboxylate
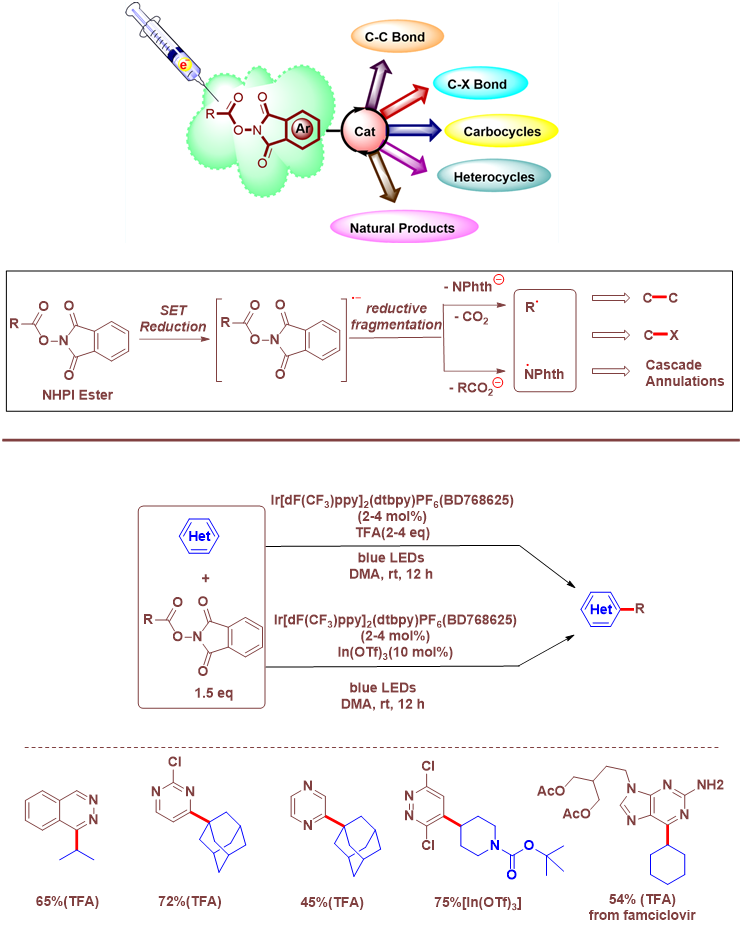
The past decade has witnessed the emergence of N-(acyloxy)phthalimides (NHPI esters) and its derivatives at the forefront of synthetic methods facilitating the construction of diverse molecular frameworks from the readily available carboxylic acid feedstock.
The NHPI esters are predisposed to undergo reductive fragmentation via a single electron transfer (SET) process under thermal, photochemical, or electrochemical conditions to generate the corresponding carbon- or nitrogencentered radicals that participate in a multitude of synthetic transformations to forge carbon−carbon and carbon−heteroatom bonds.The chemistry involving NHPI esters has received broad applicability not only in well-designed cascade annulations but also in medicinal chemistry and natural product synthesis.
Technical Notes: