BLD Insights
Enzymatic Synthesis of Glycoside - Sugar Nucleotide
14 June 2022
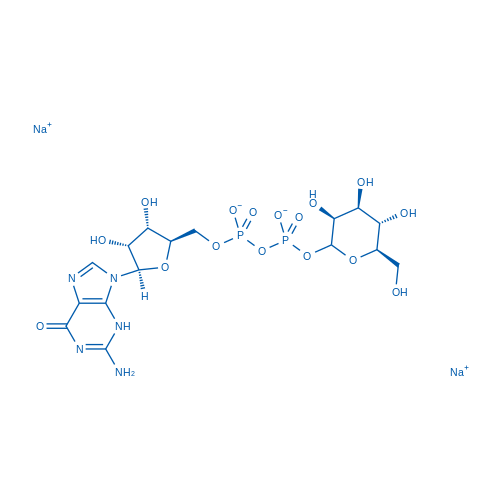
Guanosine 5'-(trihydrogen diphosphate) P'-D-mannopyranosyl ester disodium salt
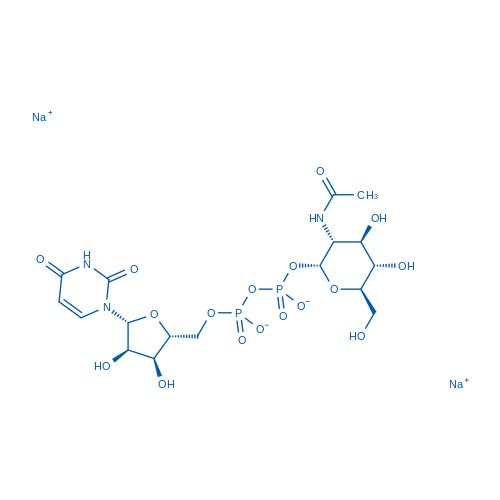
UDP-α-D-N-Acetylglucosamine Disodium Salt
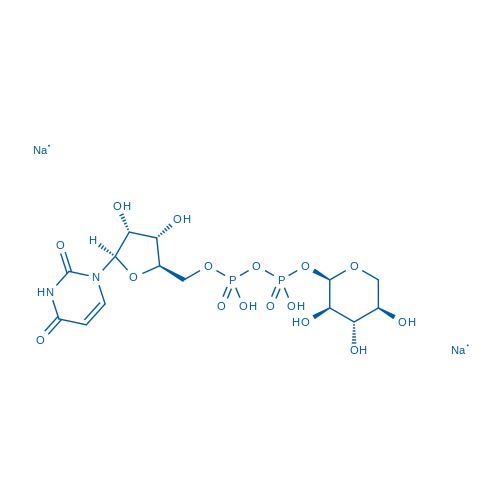
Uridine 5'-(trihydrogen diphosphate),P'-α-D-xylopyranosyl ester, disodium salt
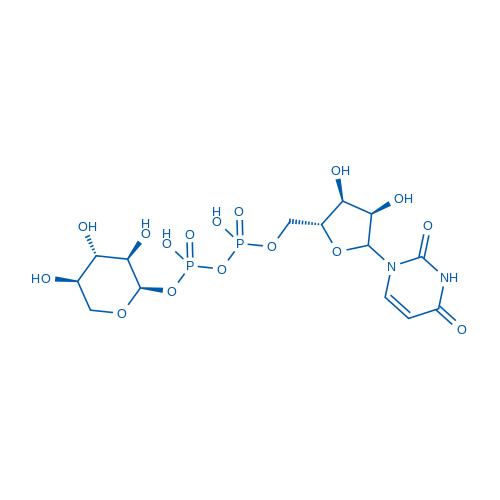
Uridine 5'-(trihydrogen diphosphate),P'-α-D-xylopyranosyl ester

Guanosine 5'-(trihydrogen diphosphate) P'-D-mannopyranosyl ester disodium salt
Glycosylation is the most crucial reaction in the synthesis of carbohydrate compounds and sugar-modified drugs. Compared to chemical glycosylation, enzymatic glycosylation can proceed stereo and regio-specifically without using protecting group manipulation procedures. Moreover, reaction conditions are extremely mild, such as the synthesis of oligosaccharide antigen Globo H (Scheme 1)1, 2.
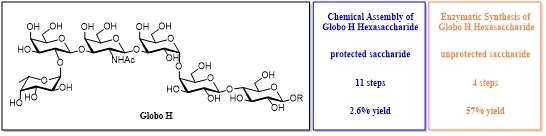
Scheme 1 Chemical and enzymatic synthesis of Globo H
The general reaction catalyzed by a glycosyltransferase summarizes the three key components (Scheme 2): (1) An acceptor oligosaccharide substrate. (2) An activated sugar donor (sugar-nucleotide) which always contains a glycosyl phosphate. This is either prepared chemically or through enzyme-recycling systems. (3) The appropriate glycosyltransferase must be available. In the past, the availability of these enzymes has severely limited their use, but as more and more glycosyltransferases are discovered and developed, enzymatic glycosylation will play a greater role.
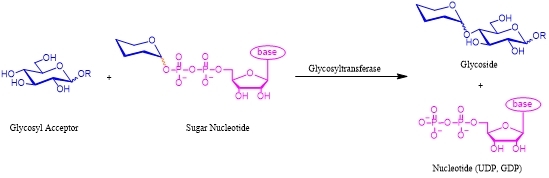
Scheme 2 Enzymatic synthesis of glycoside
As the starting material and energy driving force of the reaction, sugar nucleotides can be prepared by chemical or enzyme cycling systems. In 1994, sugar nucleotides have been produced for the first time by direct displacement of an anomeric leaving group with a nucleoside diphosphate by Hindsgaul group. The resulting anomeric mixtures of sugar nucleotides thus obtained function perfectly well in glycosyltransferase-catalyzed oligosaccharide synthesis (Scheme 3)3.
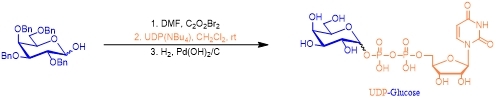
Scheme 3 Sugar nucleotide synthesized from protected sugar
In 2020, Miyagawa group developed a facile synthetic route to sugar nucleotides based on a simple condensation of unprotected sugars with uridine diphosphate using 2-chloro-1,3-dimethylimidazolinium chloride as the condensing reagent (Scheme 4)4.
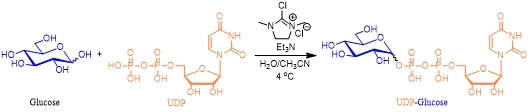
Scheme 4 Sugar nucleotide synthesized from unprotected sugar
In 2012, Linhardt and coworkers synthesized a small library of GlcNAc/GalNAc-1-phosphate analogs for their recognition by the GlmU uridyltransferase to synthesize UDP-GlcNAc/GalNAc analogues in moderate to high yield (Scheme 5)5.
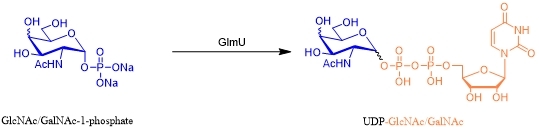
Scheme 5 Enzymatic synthesis of nucleotide
Due to its high efficiency and stereospecificity, enzymatic synthesis of glycoside has been gradually applied in the synthesis of oligosaccharides and glycoconjugates. Studies revealed that glycosphingolipids Globo H and SSEA4 were found on breast cancer cells and the cancer stem cells but were not detectable on normal cells. In 2013, Wong's team isolated and purified a variety of glycosyltransferases from Escherichia coli, combined with sugar nucleotides including UDP-Gal, UDP-GalNAc, GDP-Fuc as well as CMP-Neu5Ac, and achieved the large-scale synthesis of cancer associated antigens Globo H and SSEA4 in practical steps. These enzymatic methods were useful for the large-scale preparation of oligosaccharides for discovery research and clinical development (Scheme 4)6.
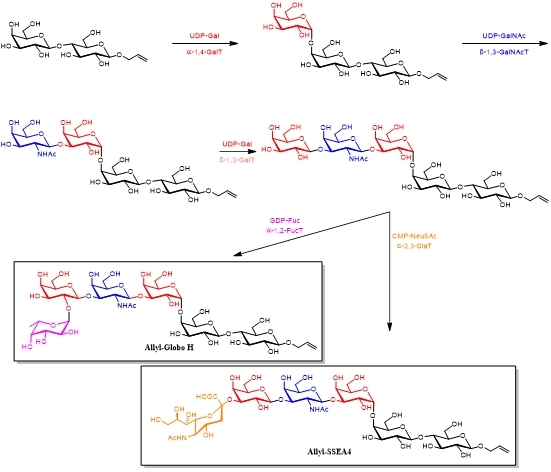
Scheme 6 Synthesis of oligosaccharides by sugar nucleotides
Glycosyltransferases and glycosidases have become essential catalysts for the construction of stereo and regio-selective glycosidic bonds in oligosaccharide structures when used in conjunction with sugar nucleotides. With the deepening of interdisciplinary research, the characteristics of specificity and high efficiency of enzyme-catalyzed glycosylation reaction, combined with computer programming technology, can realize the automatic assembly of oligosaccharides. Moreover, enzymes such as sulfotransferases, proteases, lipases, and aldolases can be exploited for the synthesis of other distinct structures which are critical to glycoconjugate function. The development of such automated systems that are easily accessible even to an unskilled researcher will have a revolutionary impact on the understanding of glycans in biological systems and on the development of carbohydrate-based therapies7-9.
References
[1]Mandal, S. S.; Liao, G.; Guo, Z. Chemical Synthesis of the Tumor-Associated Globo H Antigen. RSC Adv. 2015, 5, 23311-23319.
[2]Su, D. M.; Eguchi, H.; Yi, W.; Li, L.; Wang, P. G.; Xia, C. Enzymatic Synthesis of Tumor-Associated Carbohydrate Antigen Globo-H Hexasaccharide. Org. Lett. 2008, 10, 1009-1012.
[3]Michael, A.; Ole, H. Rapid Chemical Synthesis of Sugar Nucleotides in a Form Suitable for Enzymatic Oligosaccharide Synthesis. J. Org. Chem. 1995, 60, 14-15.
[4]Atsushi, M.; Sanami, T.; Ippei, O.; Shun, M.; Takeru, Ka.; Hatsuo, Y. One-Step Synthesis of Sugar Nucleotides. Org. Chem. 2020, 85, 15645-15651.
[5]Masuko, S.; Bera, S.; Green, D. E.; Weïwer, M.; Liu, J.; DeAngelis, P. L.; Linhardt, R. J. Chemoenzymatic Synthesis of Uridine Diphosphate-GlcNAc and Uridine Diphosphate-GalNAc Analogs for the Preparation of Unnatural Glycosaminoglycans. J. Org. Chem. 2012, 77, 1449-1456.
[6]Tsai, T.; Lee, H.; Chang, S.; Wang, C.; Tu, Y.; Lin, Y.; Hwang, D.; Wu, C.; Wong, C. Effective Sugar Nucleotide Regeneration for the Large-Scale Enzymatic Synthesis of Globo H and SSEA4. J. Am. Chem. Soc. 2013, 135, 14831-14839.
[7]Koeller, K. M.; Wong, C. Synthesis of Complex Carbohydrates and Glycoconjugates: Enzyme-Based and Programmable One-Pot Strategies. Chem. Rev. 2000, 100, 4465-4493.
[8]Koeller, K. M.; Wong, C. Complex Carbohydrate Synthesis Tools for Glycobiologists: Enzyme-based Approach and Programmable One-pot Strategies. Glycobiology 2000, 10, 1157-1169.
[9]Wen, L.; Edmunds, G.; Gibbons, C.; Zhang, J.; Gadi, M. R.; Zhu, H.; Fang, J.; Liu, X.; Kong, Y.; Wang, P. G. Toward Automated Enzymatic Synthesis of Oligosaccharides. Chem. Rev. 2018, 118, 8151-8187.