BLD Insights
Burke Boronates - Strong Backing
09 September 2022
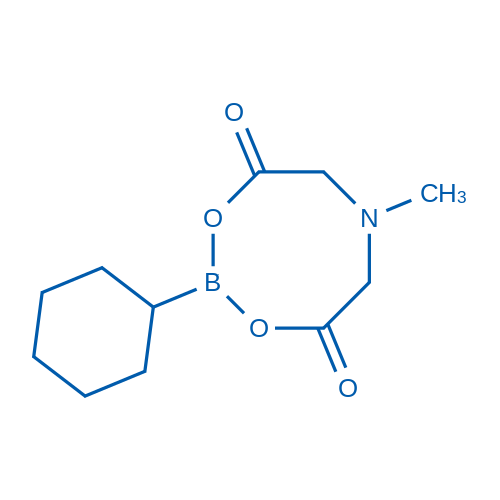
2-Cyclohexyl-6-methyl-1,3,6,2-dioxazaborocane-4,8-dione
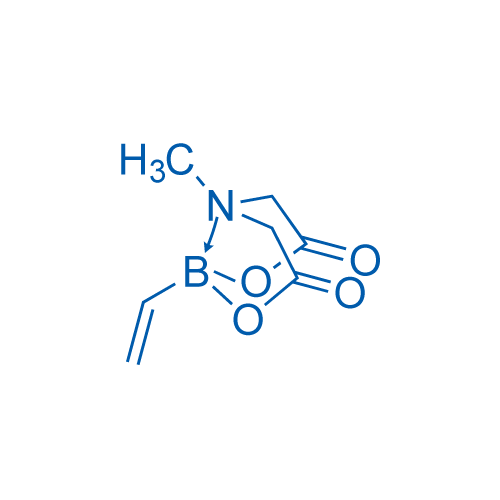
6-Methyl-2-vinyl-1,3,6,2-dioxazaborocane-4,8-dione
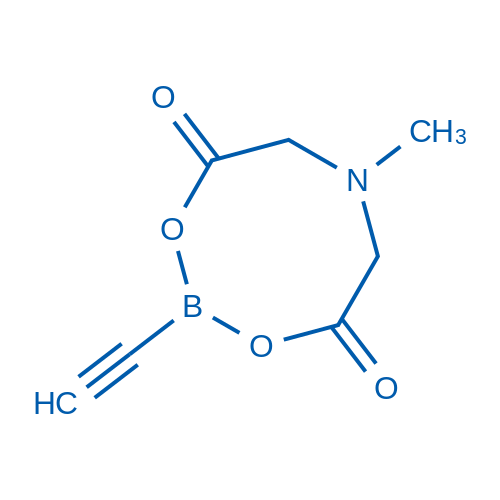
2-Ethynyl-6-methyl-1,3,6,2-dioxazaborocane-4,8-dione
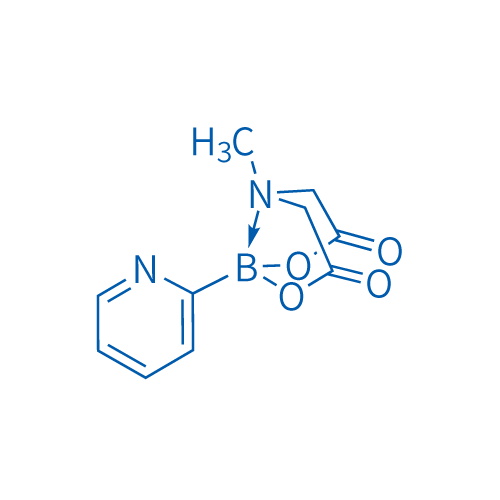
2-Pyridinylboronic acid MIDA ester
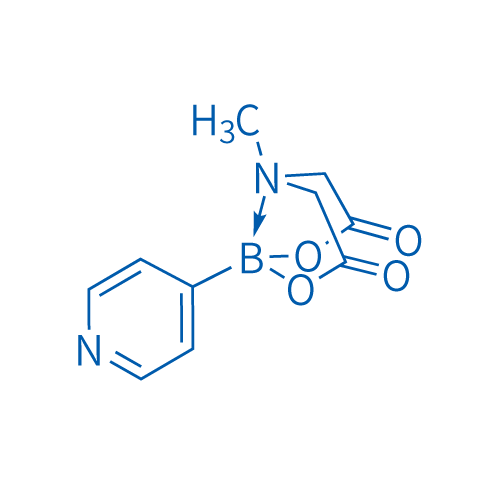
4-Pyridinylboronic acid MIDA ester

2-Cyclohexyl-6-methyl-1,3,6,2-dioxazaborocane-4,8-dione

6-Methyl-2-vinyl-1,3,6,2-dioxazaborocane-4,8-dione

2-Ethynyl-6-methyl-1,3,6,2-dioxazaborocane-4,8-dione
Burke boronates were first reported by Professor Burke in 2007 which changed the sp2 hybridization of boronic acid into sp3 hybridization and reduced their reactivity. These pyramidalized boronate esters are stable to and readily purified by silica gel chromatography (isolated as colorless crystalline solids). Halogenated Burke boronates can be conjugated first and then deprotected for subsequent conjugation, and can be iterated many times. Therefore, this reagent is widely used in the preparation of complex molecules by iterative cross conjugation.
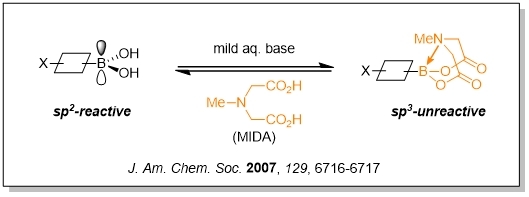
The core technology is an iterative coupling reaction assisted by Bruke borate, in which one molecular block after another reacts and joins together, much like putting Lego pieces together, greatly simplifying many complex synthesis steps and allowing automation.
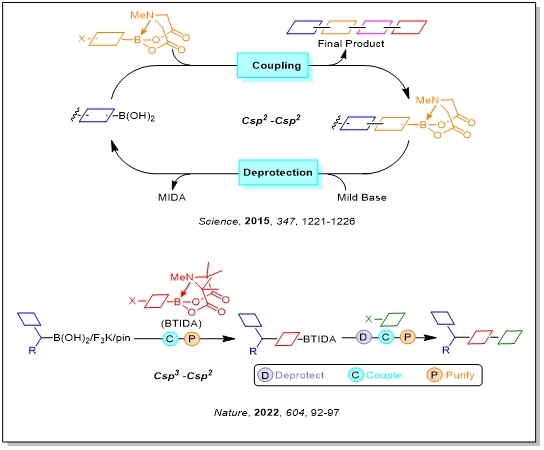
The synthesis is short and highly modular, and thus a variety of derivatives should be readily accessible simply by substituting modified building blocks into the same pathway. Further studies achieve the inherent adaptability of these methods to solid-phase and automated techniques. Although certain small molecules are at present more amenable to this approach than others, the rapidly expanding scope of the SM reaction, which increasingly includes sp3-sp3 couplings, suggests significant value for broad generality.